October 14, 2020 at 3:12 am | Updated October 14, 2020 at 3:12 am | 6 min read
Thermotolerance can be studied by the variation in photosynthesis in response to environmental factors. Chlorophyll fluorescence has emerged as an important method of determining plant photosynthetic rates, as it is non-destructive, rapid, and accurate. With the help of small portable devices, scientists have been able to find the amazing strategies plants have developed to tolerate drought and heat by tracking the various physiological processes associated with photosynthesis.
How Trees Could Cope with Climate Change
Climate change is causing temperature to increase and rainfall to decrease, making conditions arider and hotter. There is a threshold of maximum temperatures that even tree species can’t withstand; hence, global warming could be a challenge for species that occur in semi-arid regions.
The effect of climate change on trees has not received much attention. However, it is essential to know how tree species react to rising temperatures to predict their future response.
Subscribe to the CID Bio-Science Weekly article series.
By submitting this form, you are consenting to receive marketing emails from: . You can revoke your consent to receive emails at any time by using the SafeUnsubscribe® link, found at the bottom of every email. Emails are serviced by Constant Contact
In India, trees that provide timber and fodder, such as the Albizia procera, are considered essential because they are multifunctional. In central India where the tree is abundant, the continental climate has warm summers and cool winters.
Scientists decided to use the seasonal differences to measure the effect of temperature on photosynthesis, as it is known that trees show short term adjustments to climate. Thus, it could be possible to detect the effect of heat and drought stress on the trees.
Photosystem II (PSII) is heat sensitive and causes denaturation of proteins, an increase in the fluidity of the thylakoid membrane or a change in protein-lipid interaction when temperatures rise.
The scientists in India decided to measure energy use over seasons and different geographical areas of the Albizia habitats. They used reflectance indices, NDVI (Normalized Difference Vegetation Index) and PRI (Photochemical Reflectance Index), to measure non destructively with leaf spectral information.
Data was collected three times in one-year-old Albizia seedlings: during pre-winter, winter, and post-winter.
A portable photosynthesis system was used for simultaneous measurements of gas exchange and chlorophyll fluorescence, including electron transport rate (ETR). The transportation of electrons in PSII is a vital part of the photosynthesis process as it is sensitive to stress.
Spectral reflectance was measured by CI-710 Miniature Leaf Spectrometer. Reflectance at 531 nm and 570 nm were measured to calculate PRI. NDVI was derived from reflectance of infrared (800 nm) and red (680 nm) light.
The CI-710, produced by CID Bio-Science Inc., weighs less than a kilo and measures absorbance, reflectance, and transmission of light in the visible to near-infrared range. This range is useful for color analysis and measuring photochemical reactions such as photosynthesis. It gives accurate readings and can be operated in temperatures ranging from -30oC to 70oC.
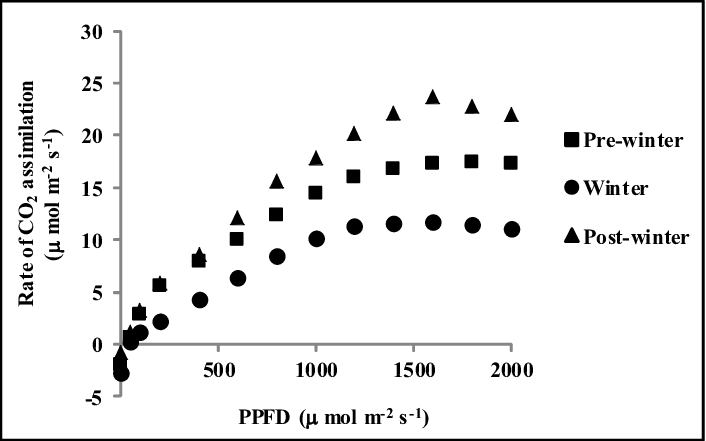
Figure 1: PPFD (Photosynthetic photon flux density) effect on CO2 assimilation of Albizia in different seasons, Alam et al. 2016. (Image credits: Indian J. of Agroforestry Vol. 18 No. 1: 96-101 (2016))
Higher canopy temperature depressions (CTD), that is the ability of the tree to lower canopy temperatures in warmer times, was observed. With CTD, plants manage to retain more of the water that they absorb. It is one of the physiological processes identified as a drought avoidance strategy, building thermotolerance capacity in plants.
Albizia had a higher carbon dioxide assimilation rate or photosynthetic rate in pre-winter and post-winter seasons, due to higher incoming photosynthetic photon flux density (PPFD). It led to more thylakoid electron transport rate (ETR) until temperatures reached 30oC.
Similarly, NDVI and PRI were higher in the warmer pre-winter and post-winter seasons than in winter.
By seeing how plants behave during winter in comparison to warmer periods, scientists were able to establish that Albizia has the inherent thermotolerance based on several physiological and cellular processes, which could help the species tackle climate change.
The study was also able to show that rising temperature effects on photosynthesis can be successfully monitored by using a combination of spectral data and vegetation indices in tropical trees.
Measuring Photosynthesis in CAM Plants
There are many ways of measuring the photosynthetic rate of plants. In desert plants, the usual method of measuring gas exchanges presents a challenge due to the thick fleshy tissue of the succulents and their Crassulacean Acid Metabolism (CAM).
To prevent loss of water through evapotranspiration during the high daytime temperatures in the desert, CAM plants keep their stomata closed during the day and open them in the evening. These plants absorb the carbon dioxide needed for photosynthesis during the night and store it as malate.
Since the stomata of CAM plants are closed when photosynthesis occurs during the day, the normal gas exchange measuring tools are not useful.
In such scenarios, measuring chlorophyll fluorescence and electronic transfer rate (ETR) are viable alternatives, as they also provide information on the influence the environment has on photosynthesis.
To do so, scientists have to factor in the various morphological adaptations that plants have developed to remain cool, such as the thick waxy layers on the leaf surface that make them lighter in color or the presence of a high density of hairs or spines. These measures help the leaves reflect light and prevent the leaves from getting warmer.
However, this can affect the amount of light that the leaves absorb, and scientists cannot use a standard value for absorbance (α) to estimate ETR, as it can lead to inaccuracy.
Hence, scientists in California studied 56 CAM species to find out the variation of absorbance (α) in succulent plants and how this effects ETR. They also collected data from two species with usual C3 photosynthesis pathways: one with flat leaves, and the other with hairy leaves for comparison.
PSII efficiency or chlorophyll fluorescence was measured by a pulse amplitude modulated fluorometer at 1,980 μmol m–2 s–1, based on which ETR was calculated.
Immediately after, reflectance and transmission at PAR (400–700 nm) were measured at the same site on the leaves to calculate absorbance values. The instruments used in the measurement are now out of production, but the scientists recommended leaf spectrometers such as CI-710s SpectraVue Leaf Spectrometer manufactured by CID BioSciences Inc.
The CI-710 can simultaneously calculate absorbance, transmittance, and reflectance in the visible and near-infrared range of light. It is a small, portable device that can also quantify film thickness.
The scientist found a wide variation in absorbance, from 0.55 to 0.92, among the 56 species, but the mean for all species was an α of 0.84, similar to C3 plants. However, most CAM species tend to have a lower α value.
The scientists then calculated ETR at the mean absorbance (0.84) and measured absorbance for each species, finding a significant difference. For species with absorbance close to 0.84, there was not much difference.
However, in species with reflective leaf hairs, such as in Encelia farinosa, the ETR calculated with actual absorbance was 53% lower than ETR at mean absorbance (0.84). Hence, when a species has light-colored or reflective photosynthetic tissue, it is important to use the absorbance value of each species to calculate their ETR and not any standard average absorbance value.
The scientists suggest creating a database of light absorbance values for all species that can be used when measuring ETR or other photosynthetic parameters.
Several Responses to Thermotolerance
A single parameter, such as electron transfer rate estimated by chlorophyll fluorescence, can track the connection between spectral traits, photosynthesis, and intercellular biochemical reactions. While some use it to study the short-term effects of heat stress, others probe the basic thermotolerance metabolism of plants to survive in extreme climates through long-term adaptations.
—
—
Vijayalaxmi Kinhal
Science Writer, CID Bio-Science
Ph.D. Ecology and Environmental Science, B.Sc Agriculture
Feature image courtesy of Josef Wells
Sources
Alam, B., Newaj, R., Rizvi, R.H., Prasad, R., Handa, A.K., Chaturvedi, O.P., Chaturvedi, M., & Singh, A.K. (2016). Characterizing the thermotolerance properties of Albizia procera in temporal and seasonal scale through physio-biochemical traits and spectral indices. Indian J. of Agroforestry, 18 (1), 96-101.
Editors, B. (2019, March 27). CAM Plants. Retrieved August 26, 2020, from https://biologydictionary.net/cam-plants/
Stemke, J.A., & Santiago, L.S. (2011). Consequences of light absorptance in calculating electron transport rate of desert and succulent plants. Photosynthetica 49, 195–200. https://doi.org/10.1007/s11099-011-0026-y
Kumar, S., Singhal, T., Bharadwaj, C., Kumar, T., Singh, M.P., Tripathi, S., Roorkiwal, M., & Varshney, R. (2016). Canopy temperature depression (CTD) as a measure for Drought Resistance in a training population in chickpea. Conference: National Seminar on Omic Technologies for better food and nutrition: Department of Biotechnology, Telangana University, Nizamabad, Telangana, India. Retrieved from https://www.researchgate.net/publication/301560609_Canopy_temperature_Depression_CTD_as_a_measure_for_Drought_Resistance_in_a_training_population_in_chickpea
Related Products
Most Popular Articles
- Transpiration in Plants: Its Importance and Applications
- Leaf Area – How & Why Measuring Leaf Area…
- How to Analyze Photosynthesis in Plants: Methods and Tools
- Forest & Plant Canopy Analysis – Tools…
- The Forest Canopy: Structure, Roles & Measurement
- The Importance of Leaf Area Index (LAI) in…
- Root Respiration: Importance and Applications
- Stomatal Conductance: Functions, Measurement, and…
- Irrigating with Saline or Seawater
- Crop Water Use Efficiency Explained




